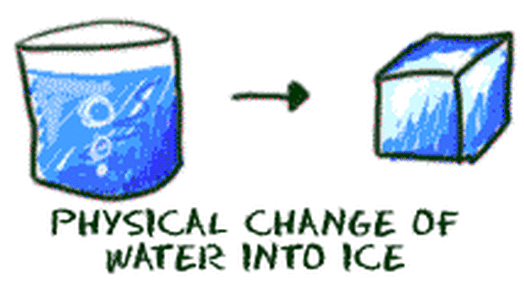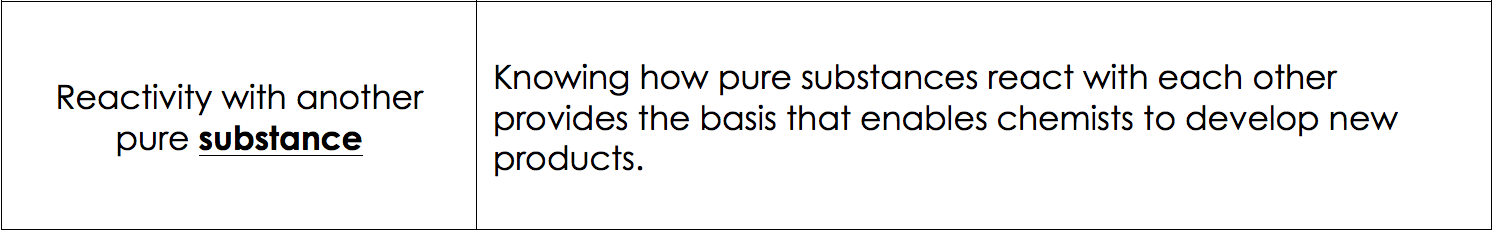Lesson 2:
How do we use properties to help us describe matter?
Learning Goals:
1. To learn the difference between Physical and Chemical properties.
2. To learn the types of Physical and Chemical properties.
3. To understand the difference between Qualitative and Quantitative.
4. To understand what we mean by Reactivity.
2. To learn the types of Physical and Chemical properties.
3. To understand the difference between Qualitative and Quantitative.
4. To understand what we mean by Reactivity.
Chemistry IS cool: PART 2 OF ∞
What are Properties?
Physical Properties:
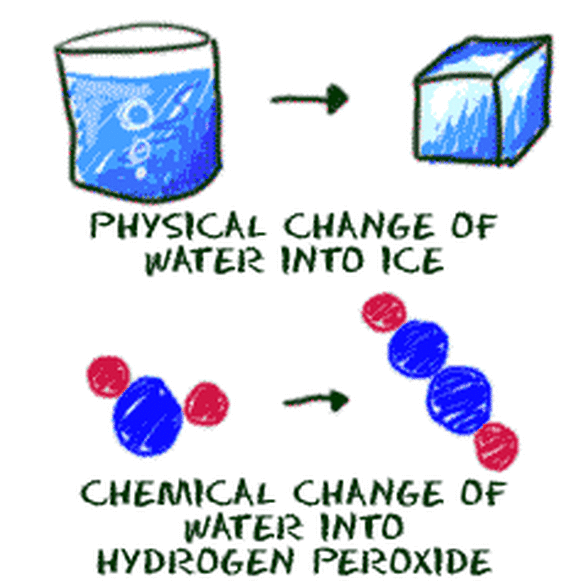
Physical Properties = Characteristics that don't change the substance.
Physical Change = NO NEW substance is formed!
Physical Change = NO NEW substance is formed!
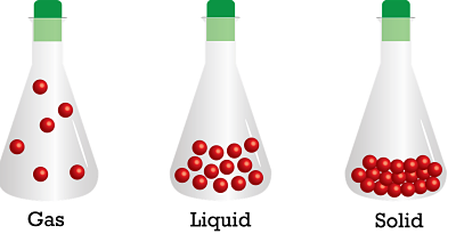
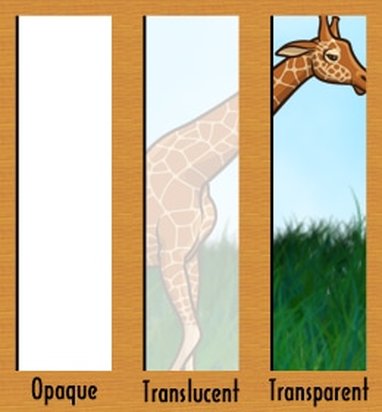
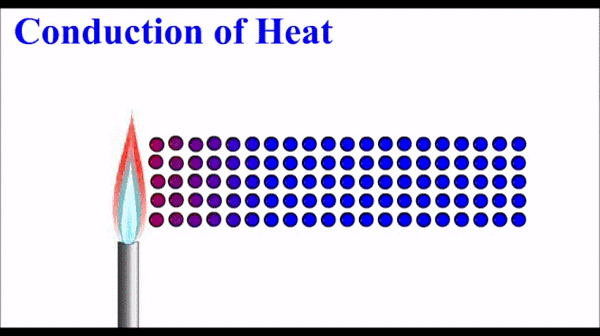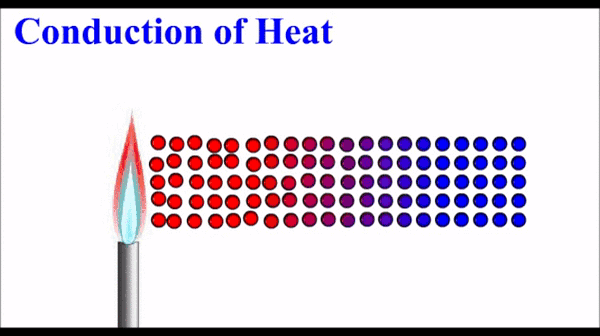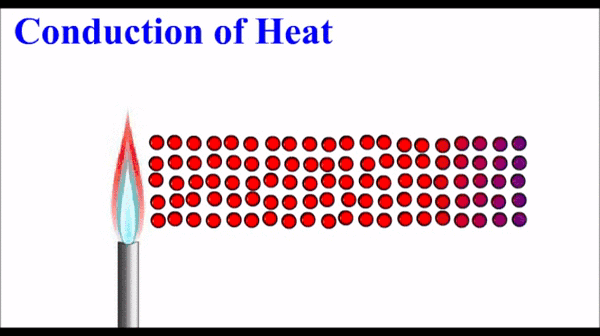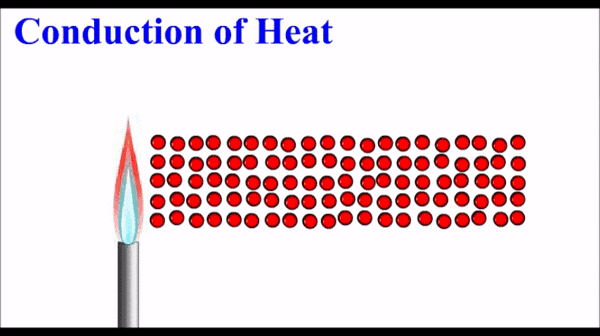
How to Describe Matter Physically:
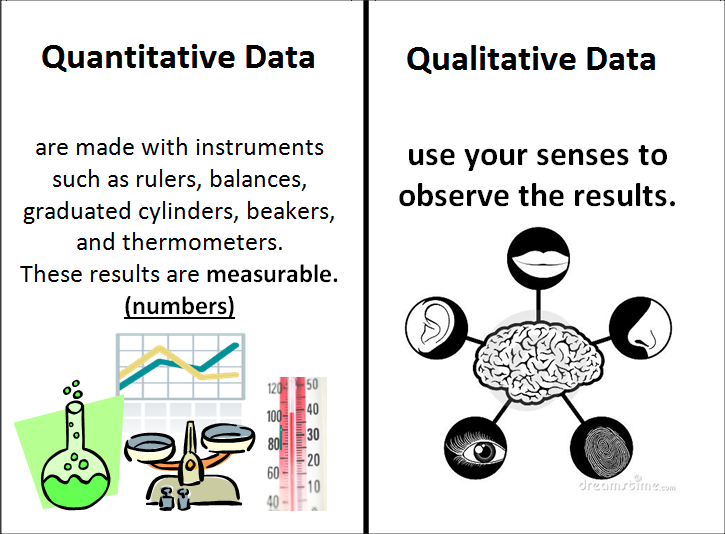
(Qualitative & Quantitative Observations)
(Qualitative & Quantitative Observations)
Chemistry challenge:
Which physical property is it?
Chemical Properties:
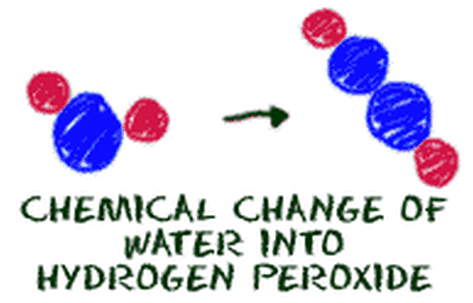
Chemical Properties = Characteristics that CHANGE the substance.
Chemical Change = a NEW substance is formed!
Chemical Change = a NEW substance is formed!
Reactivity with Other Substances:
Other Chemical Properties of Matter are:
Chemistry challenge:
Which Chemical property is it?
QUICK Review:
|
Physical Properties:
Features that can be observed without changing type of substance.
Physical Change:
NO NEW substance is formed!
|
Chemical Properties:
Features that can be observed only by changing the type of substance.
Chemical Change:
A NEW substance is formed!
|
Quizlet: Lesson 2
Learn about Physical & Chemical Properties on Quizlet!
HOMEWORK: