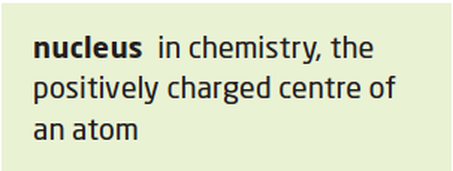Lesson 3:
Evolutions of the atomic model
Learning Goals:
1. To learn how the Atomic Model evolved throughout the years.
2. To understand how and why scientists came up with their own theories of what the atom looks like based on experimentation.
2. To understand how and why scientists came up with their own theories of what the atom looks like based on experimentation.
Chemistry IS cool: PART 3 OF ∞
Activity #1: Paper Tear-Off!
1. Grab a piece of paper.
2. Tear the paper in half.
3. Do this again and again until you can't tear anymore.
4. Count how many tears you could get to in total.
2. Tear the paper in half.
3. Do this again and again until you can't tear anymore.
4. Count how many tears you could get to in total.
Activity #2: Mystery Box of Science!
1. Each person will get 10 seconds to shake the mystery box of science.
2. At the end, each person will get one guess at what is inside the box.
3. If you get it right, you are a freakin' genius!!!
2. At the end, each person will get one guess at what is inside the box.
3. If you get it right, you are a freakin' genius!!!
Activity #3: Evolution of the Atom
1. Use the textbook notes below to learn about the following scientists and their atomic theories:
Dalton
Thomson
Rutherford
Bohr
Chadwick
Thomson
Rutherford
Bohr
Chadwick
2. Answer the follow-up questions in your class notes as you read the chapter.
Textbook Notes: Chapter 5.1
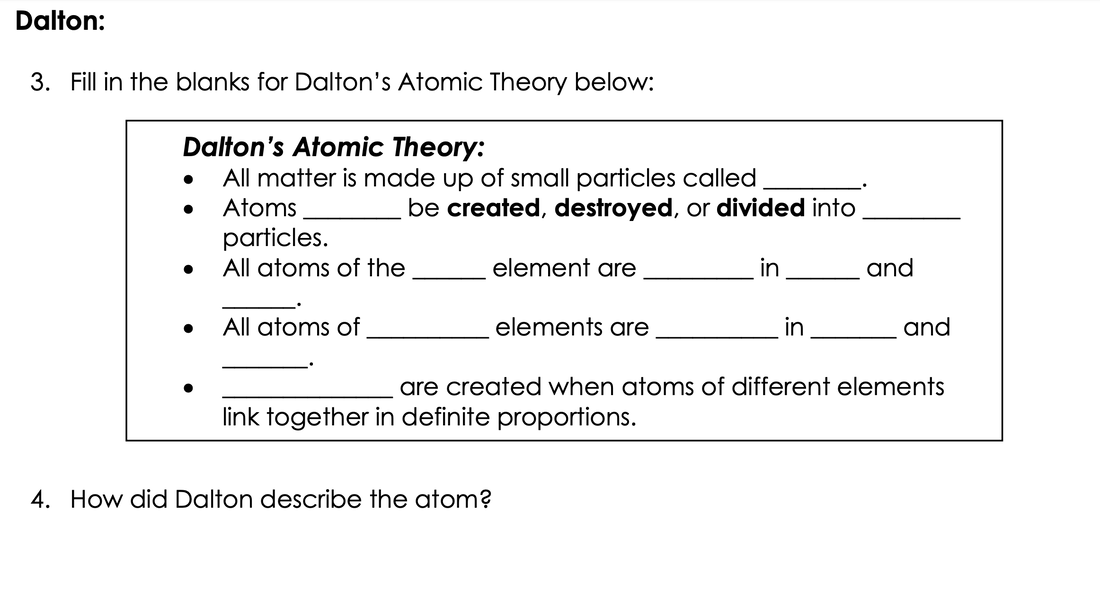
Dalton:
Thomson:
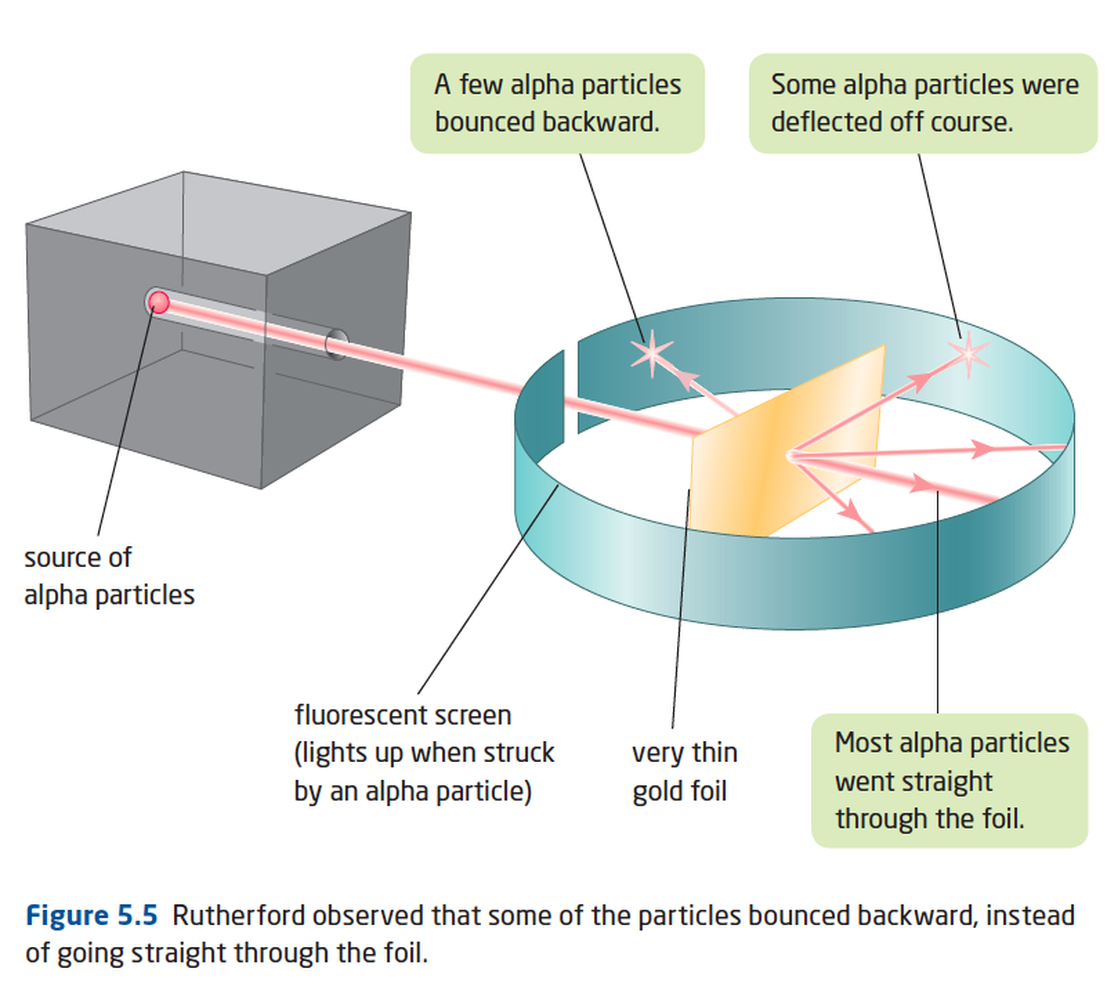
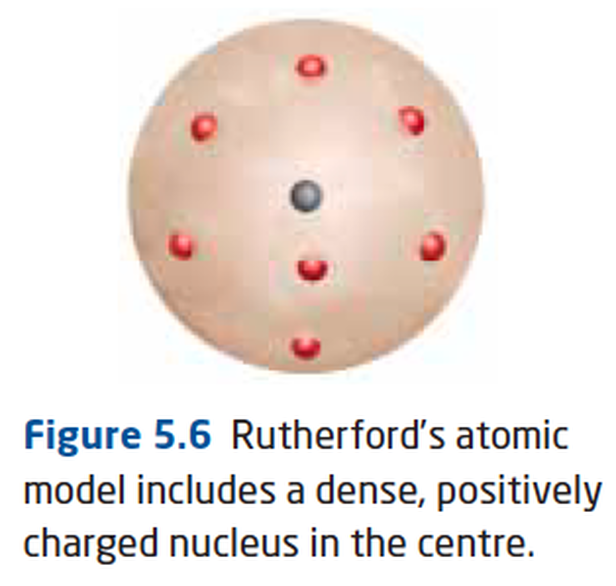
rutherford:
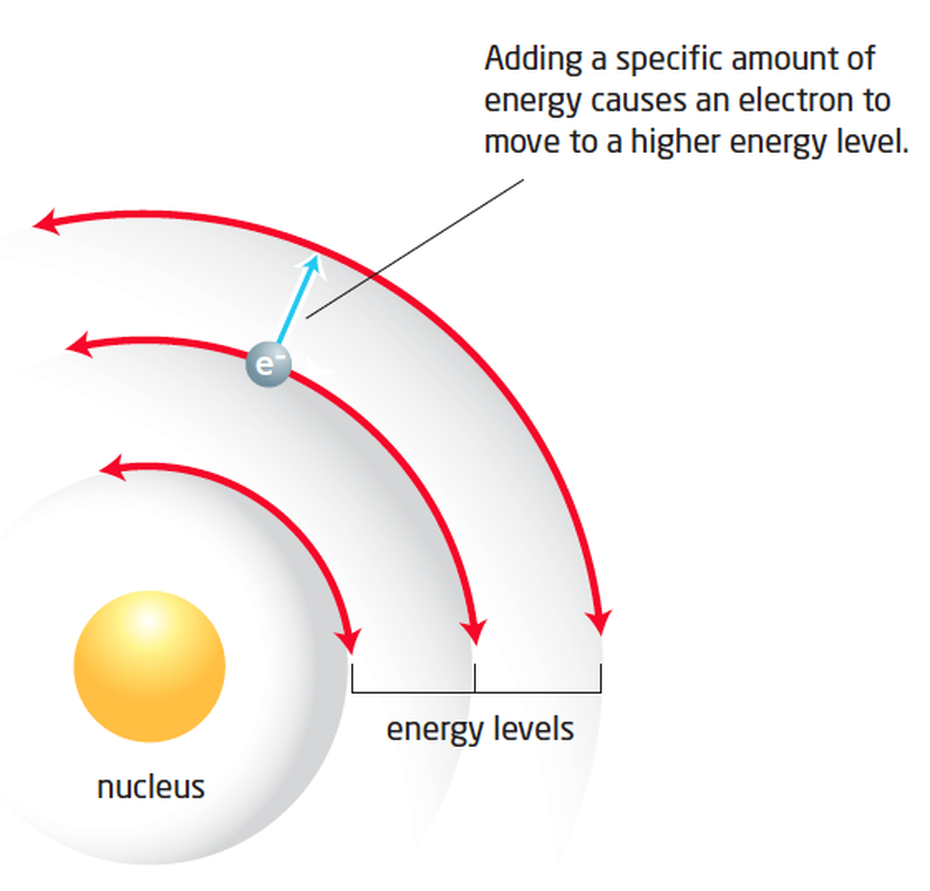
Bohr:
Chadwick:
Quizlet: Lesson 3
Learn all about Evolutions of the Atomic Model on Quizlet!
HOMEWORK: